Simplifying disease testing and reporting for under-resourced sites
Summary
During the COVID-19 pandemic, public health departments faced the challenge of collecting diagnostic test results from an unprecedented number of new data sources, including non-traditional testing facilities like K-12 schools and nursing homes — many with no prior experience in public health reporting. We worked with the Centers for Disease Control and Prevention (CDC) to build and launch a time-saving tool that’s made it easy for over 17,000 of these facilities to record and report over 8 million test results since November 2020. As we continue supporting CDC’s COVID-19 reporting efforts, we’re expanding this tool to streamline reporting for other infectious diseases.
The challenge
To respond to disease outbreaks quickly, public health departments need complete, reliable, and actionable disease incidence data. For example, during the COVID-19 pandemic, the test positivity rate informed policy interventions, such as mask guidance, stay-at-home orders, and limits on indoor activities. Simply put — better data enables public health departments to make informed decisions quicker, which helps all of us.
At the height of the COVID-19 pandemic, testing expanded outside of traditional health settings to facilities such as K-12 schools and retirement communities. However, collecting and sending accurate testing data in these settings was difficult — if it happened at all. Non-traditional point-of-care testing facilities typically lacked IT infrastructure to send data to public health departments. Many recorded test results on paper then faxed them to the appropriate department, a time-consuming process that was usually outside a test administrator’s primary job. Patients were also less likely to provide demographic information in these unconventional settings, leading to incomplete and lower-quality data for health departments.
Adding to the complexity were health departments’ differing requirements. As one interviewee who reports to multiple jurisdictions noted:
It’s a challenge to do the test, put it on paper, then go back and enter it in both formats — for the county and state.
Even when facilities did have tools to track patients and results, many lacked the ability to automatically report to their jurisdictions in the different formats that they required. On the receiving end, public health departments had to process manual reports sent from a variety of locations, which took time and resulted in errors. This manual and cumbersome process made it difficult to act fast and stay informed.
The solution
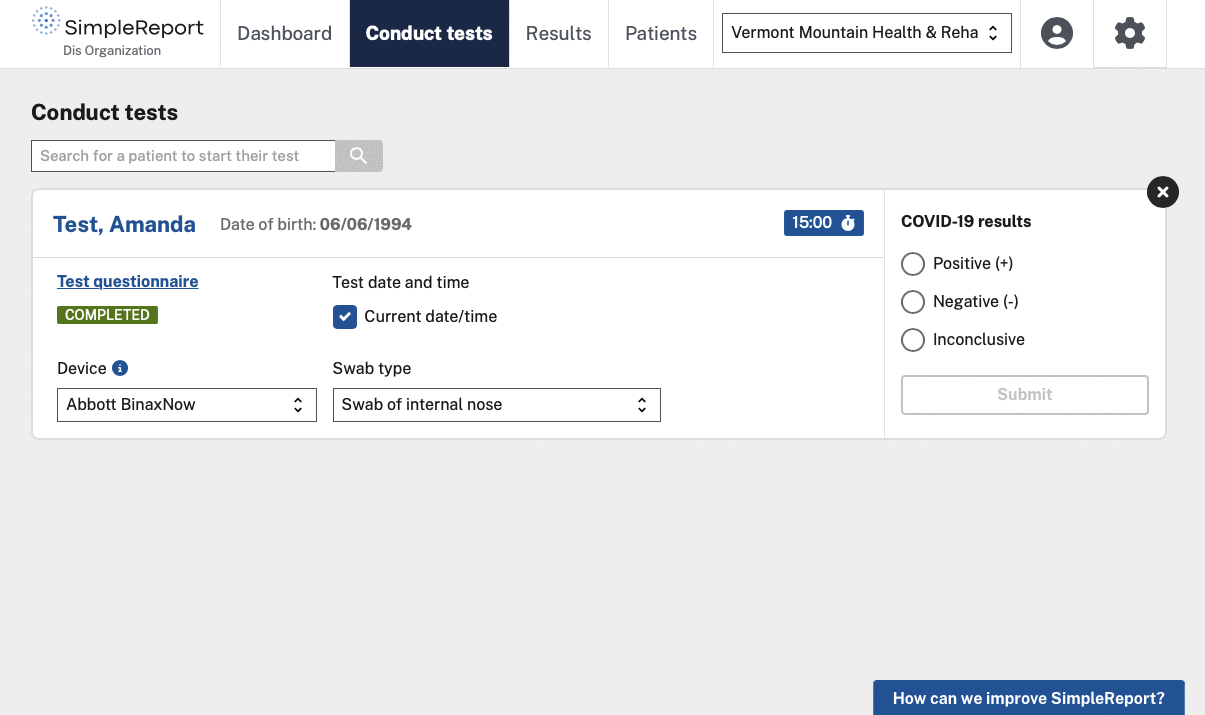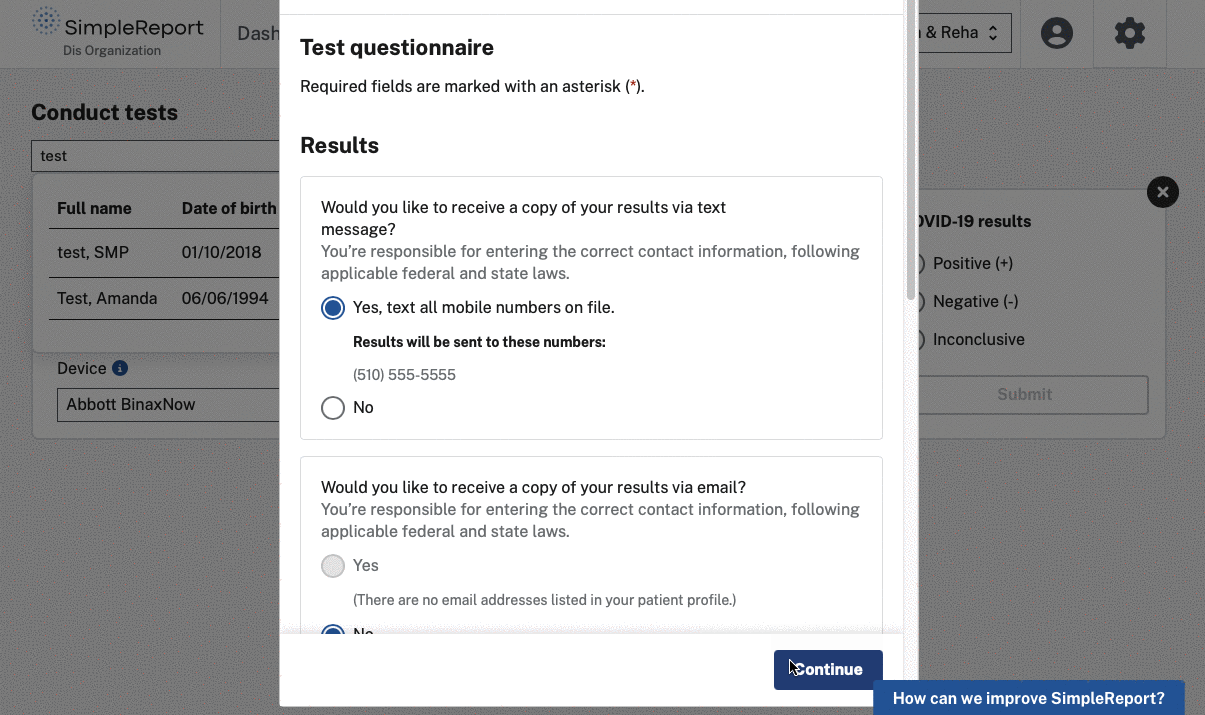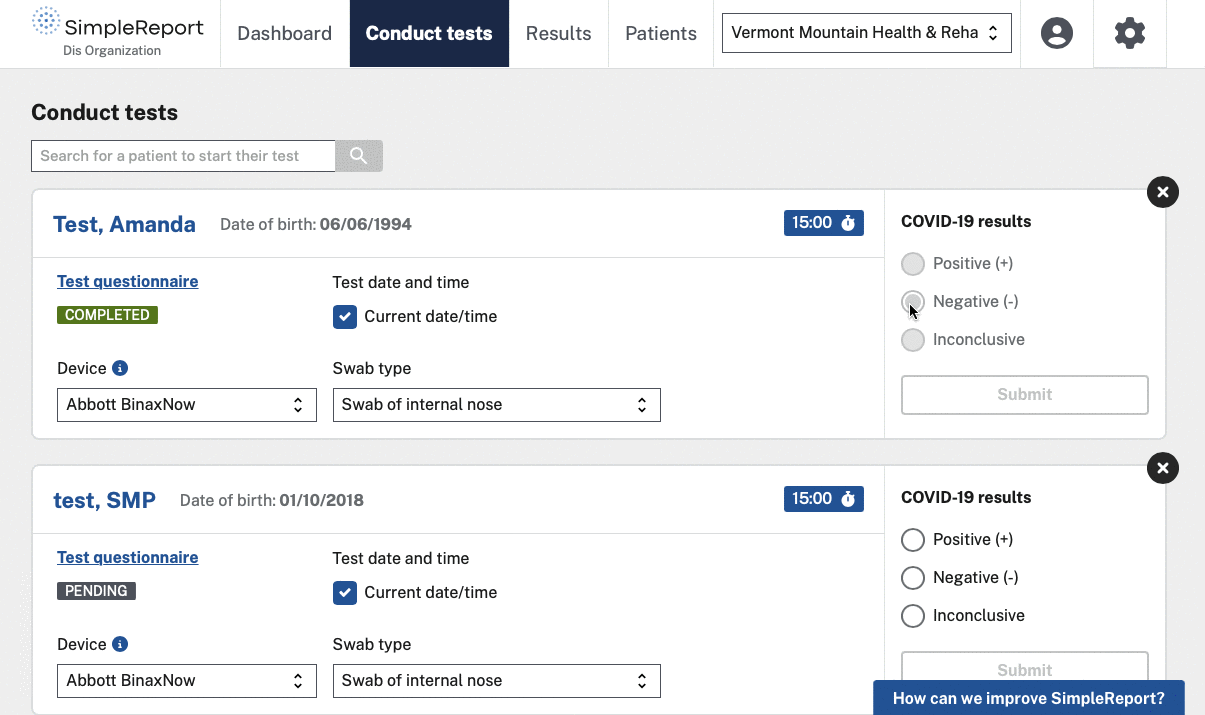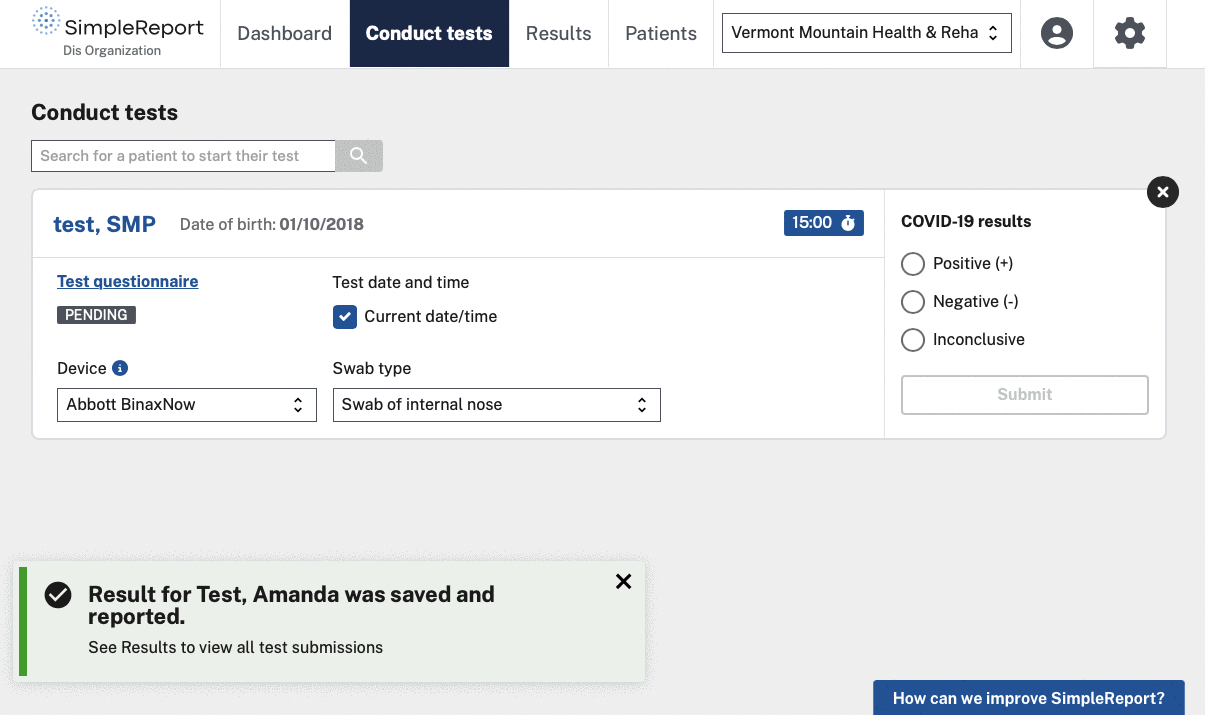
We helped CDC and the U.S. Digital Service (USDS, now U.S. DOGE Service) launch and grow SimpleReport, a free tool that makes it easy for non-traditional testing sites to record rapid test results and report them to public health departments. The tool serves as both a workflow and reporting platform — sites use it to manage patients and results, and SimpleReport automatically sends structured data to health departments via ReportStream in whatever format and cadence each jurisdiction requires. No fax machines, no manual data entry, no duplicate reporting.
Going from what we were doing to this is awesome. It’s very easy to use.
Once we assumed full management of SimpleReport from our USDS partners, the focus shifted to adoption at scale. We redesigned the account creation, login, and password reset experiences to speed up onboarding while maintaining security. We added bulk CSV upload so facilities could report large volumes of results without entering them one at a time. And we built an analytics dashboard and one-click results download to reduce reporting burden beyond what goes to public health departments.
Scaling also meant keeping the platform reliable for a growing user base. Automated service monitoring, continuous deployment, and 24/7 on-call support kept issues from reaching end users — when a feature’s code passes testing, it’s live for users within 20 minutes. We also integrated accessibility reviews into design and engineering workflows to maintain Web Content Accessibility Guidelines (WCAG) Level AA compliance throughout.
With the COVID-19 foundation in place, the team turned to a bigger question: could SimpleReport work for other diseases? We added support for multiplex devices that test for both COVID-19 and flu, then partnered with California’s Department of Public Health to launch flu and respiratory syncytial virus (RSV) reporting for facilities that previously had no way to report electronically. We also established a pilot with the Los Angeles County Department of Public Health to support HIV and STI reporting — proving that the tool’s modular design could adapt to new diseases without rebuilding the core platform.
The result is public health reporting infrastructure that didn’t exist before — connecting thousands of facilities that were invisible to surveillance systems and giving them a path to report on whatever comes next. SimpleReport isn’t just a COVID-era tool; it’s the structural groundwork for faster, more complete responses to future health crises.
The results
- Over 17,300 facilities reporting tests through SimpleReport across all 50 states, as well as other U.S. territories and Canadian provinces
- Over 8.2 million tests reported since launch (from January 2021 to April 2024)
- More than 2.3 million text messages sent notifying patients of their test results
- Cited as a Data Modernization Initiative (DMI) success story in a presentation to CDC Director Dr. Walensky for its “game-changing” work in Alaska
- Support for flu reporting launched in July 2023 and RSV reporting went live in January 2024 following a successful pilot with California
- More than 1,150 facilities in five states have joined the flu and RSV pilot, reporting over 143,000 flu test results and over 4,200 RSV test results
- Support for HIV reporting is scheduled to go live in April 2024 with the Los Angeles Community Health Project